News of Friends of Grasslands
Supporting native grassy ecosystems
July - August 2025
ISSN 1832-6315
Also available as a pdf file (2.6 MB) in original format with photos
In this issue
Acid Grasslands: another perspective
Fungi: recyclers, decomposers, parasites, symbiotic partners and more
Three ‘natives’: close-up
Hiding in the grass: uncovering Melichru (Ericaceae) species diversity in grassy ecosystems
The Mulloon Institute, and a search for NTG
Activities
Work Parties
Gurubung Dhaura (Stirling Park):
- Sat 12 July 9-12:30pm. Vinca control, Register Andrew Zelnik
- Sat 9th Aug 9-12:30pm. Vinca control, Register: Jamie Pittock
Budjan Galindji (Franklin Reserve)
- Wed 2 & 23 July 9-11:30am, Register: Margaret Ning
- Wed 6 & 27 Aug 9-11:30am, Register: Margaret Ning
Save the date
- Sat 23 Aug 1.30 to 4.30pm for our mid-winter event at St Mark’s. Theme: ecosystem restoration - speakers Ben Serafin from South East Local Land Services, Brian Butler Kemp and one other from ACT NRM, and FOG member Ken Hodgkinson of CSIRO. Register: Margaret Ning
New members
- From 1 Nov 2024 to 31 May 2025 10 new members: 7 ACT; 1 VIC; 2 NSW. A very big welcome to all of you!
President’s report
Matt Whitting, Vice-President, Friends of Grasslands
A review: Close to the Edge
In June 2024, the ACT Government Response to the ACT State of the Environment Report 2023 (available here, page 2; the original report is available here) said “it is a sad and unfortunate reality that pressures on the environment continue to grow and evolve”. This response appears to have lit a fire in the belly of the then ACT Commissioner for Sustainability and the Environment, Dr Sophie Lewis.
In May 2025, her report Close to the Edge: An Investigation into the Effects of Urban Expansion on the Environment of the ACT (Report available here) was tabled in the Legislative Assembly. It details the impact of 20 years of urban growth on biodiversity in the ACT. The Report explains how and the extent to which threatened ecological communities have declined in extent and condition over the period, particularly Box-Gum Grassy Woodland and Natural Temperate Grassland (‘grassy ecosystems’).
The Report includes a short section from Jamie Pittock: "Grassy ecosystems are among the most endangered in Australia and they are home to many of the ACT’s most endangered species. Here (on pages 116-119) the challenges and opportunities to conserve these ecosystems and benefit Canberrans as our city develops are outlined."
It states that the ACT’s urban footprint expanded by 40 per cent between 2004 and 2023 largely due to greenfield developments which have led to direct habitat loss, fragmentation, and degradation. Key threats that have affected ecological communities have included mature tree loss, disruption of habitat connectivity and ongoing disturbances such as weed encroachment, unrestrained pets and light and noise pollution. Despite existing conservation policies, Dr Lewis found biodiversity is not sufficiently prioritised in legislation and policy implementation, leading to continued environmental degradation.
Relevantly, the Assembly voted on 10 April 2025 to define an urban growth boundary by June 2027 (item 4 in Notice 2 on pp. 206-207 available here).
This could help, literally, draw the line. However, doing that raises the question of whether the high conservation value (HCV) sites, mature native trees and biodiversity corridors within this boundary will be adequately conserved. Work Sarah Sharp is leading to identify and map HCV sites outside the ACT’s protected reserves is enabling FOG to better advocate both the establishment and the more-coordinated management of a biodiversity network that will, we hope, connect and conserve all the ACT’s HCV grassy ecosystems.
Like Dr Lewis, it is clear FOG members do not accept that the ACT’s nature must be sacrificed in the pursuit of relentless growth. Thank you to all who volunteer to ensure Canberra’s expansion is cautious and respectful. Whether you do so by maintaining a grassy ecosystem patch, documenting the habits of a weed or writing to a government minister about your priorities, your time and effort matters. Where they are unsubstantiated, we should continue to reject promises of so-called ‘nature positive’ development.
Within a few months, the ACT community will be invited to comment on a draft of the next ACT nature conservation strategy. It is expected to identify what work programs will be prioritised, and to set targets. May I encourage you to comment on it! Dr Lewis’ report demonstrates this next strategy could not be more important.
[Editor’s note: President Jamie Pittock is working overseas during July and August]
Acid Grassland - another perspective
Margaret Ning
It is an email from FOG member Matt Mullaney, who recently visited Kew Gardens in London, that has recently added to my ‘grassland’ knowledge. I was aware of tall prairie grassland and meadow grassland in the US, and of course the savannah in Africa that has our signature ‘grassland species in common’ (Themeda), but I hadn’t heard of ‘acid grassland’ before.
This signage at Kew Gardens (photo) looks similar to what we erect here in Australia to alert the public to areas of significant vegetation, taking the opportunity to introduce them to the ecosystem in general as well as some of its significant species. My text boxes below provide the text of what is shown on the sign.
Acid grassland
The natural area is home to some of the last fragments of acid grassland that exist in Kew Gardens.
Acid grasslands are usually formed on free draining, acidic soils that are low in nutrients. Lowland acid grassland has been made one of the priority habitats to protect by The London Biodiversity Action Plan (BAP).
Ant hills, like the ones you see in the centre of the meadow, are a common feature of acid grassland. This habitat attracts a specific group of insects named the ‘Thames Terrace Invertebrates’. Along with the ants and molluscs, many solitary insects create small burrows in the soil. They attract a variety of birds to Kew, including green woodpeckers, meadow pipits and song thrushes.
'By the middle of spring there may be little heaps of sandy material surrounding a miniature crater which leads to a deep hole, like the pipe of a toy volcano ... shortly a red tailed bee approaches, goes down the shaft, performs its business, and departs’.
Walter Johnson,‘Animal Life in London’, 1930
But it was the acid grassland species on the signage that gave Matt and me a laugh. Pictures of cat’s ear (Hypochaeris radicata), Common Stork’s Bill (Erodium cicutarium) and Sheep’s Sorrel (Rumex acetosella), reminded us of three of our common local weeds, and Matt’s images taken in the remnant acid grassland area also contained some of our local target weeds, especially Fog Grass (Holcus lanatus).
Matt’s comment on the Kew signage: “It is interesting to note that the same grasslands they consider to be beautiful native grasslands looked nearly identical to the exotic grasslands of Canberra such that I wouldn't be able to tell you what country I was in, based on the flora alone”.
The photos below show the acid grasslands - desirable plants there and weeds here (reference here).
All photos in this article are by Matt Mullaney
Fungi: recyclers, decomposers, parasites, symbiotic partners and more
Michael Bedingfield
Fungi are an amazing form of life and perform a number of roles in the environment. Most of the time they are not visible. What we see is the fruit body, which is the spore producing structure of the fungus. From the appearance of the fruit body we get the names for the various kinds of fungi. What we don’t see are the long, branching, filamentous structures that grow through and within the soil, wood, animal remains or other material. A lot of what fungi do is therefore hidden from view but very important in many ways.
Fungi feed by absorbing nutrients from the environment around them, taking in carbon compounds, nitrogen and other organic chemicals. The filamentous structures of fungi are called hyphae and they form into a complex, many-branched network. Collectively this structure is called the mycelium. Hyphae secrete digestive enzymes into their surroundings causing those materials to break down making it possible for the fungi to absorb the nutrients therein. That is, they digest their food externally, and then absorb the soluble nutrients. In this way they decompose decaying plant and animal tissue thus acting as recyclers for these organic materials. By fungal activity solid material such as wood can gradually disintegrate and disappear into the soil. Because of the way fungi are able to convert organic matter into nutrients in the soil, plants are able to use those nutrients to grow.
Some fungi may act as parasites, growing and feeding on a living host. They use their digestive enzymes to break down living tissue. This may cause illness for the host. The Chytrid fungus Batrachochytrium dendrobatidis is an example. It affects amphibians and is a worldwide problem. It has caused a decline in the number of frogs and the extinction of some species.
Other fungi have symbiotic relationships with other organisms. Most mycorrhizal fungi have a mutually beneficial relationship with plants and are found everywhere in the soil. Their symbiotic relationship means that particular fungi and their host plants benefit from each other and also depend on each other for their survival. Also neither is worse off for the relationship. For example, many orchids have a special relationship with mycorrhizal fungi, some orchids being dependent on one or a small few species of the fungi.
Orchid seeds are tiny, like specks of dust, and don’t contain within them enough nutrients to produce a new shoot. But with the help of the mycorrhizal fungi they get all the food requirements to grow. In Australia these fungi are essential for many terrestrial orchids for seed germination, nutrient uptake and general development, especially in the early stages of life. Fungi are unable to fix carbon. But later on in the life of the orchids, through photosynthesis, they provide their allied fungi with carbohydrates they need.
More than a few native orchids are threatened species. So understanding the mycorrhizal fungi and their associations with particular orchids is important for their conservation.
Coralloid fungi are characterized by their highly-branched fruit bodies, which look very much like corals. I have provided a photo of a specimen taken from above (left).
It is yellowish in colour and of the genus Ramaria and is very attractive. Coralloid fungi can have a range of colours other than my example including white, blue, purple, orange and red. Most species grow on the ground but some also grow on decaying wood. The specimen was photographed at Six Mile TSR near Bungendore.
Phaeotrametes decipiens is a polypore fungus. My photograph shows it as being reddish dark brown, growing on a tree. Because of its appearance it is called a bracket fungus or shelf fungus.
This species grows on dead wood and is fairly tough itself. It grows out in concentric circles which are different shades of brown.
The upper surface is a bit bristly to the touch when fresh, smoother when old. The underside is pored or maze-like, looking a bit like a honeycomb. The polypore in my photo was found on Tuggeranong Hill.
Another type of fungi is the earthstars. My example is from the genus Geastrum. The fruit body of earthstars is a puffball-shaped spore sac sitting on a star-like base.
The example in my photograph has aged a bit, and the petal-like star-shaped base has curled downwards. The spore sac is pointed at the top and this is the opening through which the spores puff out when the fruit body is old and compressed in some way.
The immature fruit body is the spore sac within a thick outer casing. The outer casing splits when it is mature and folds outwards to create the star shape. Earthstars are usually found growing in soil, in a variety of places including among leaf litter in woodlands or forest. The specimen in the photo was growing in my garden.
The fruit bodies of fungi can have many weird and wonderful shapes and I have shown just a few of them. The field guides on fungi provided by Canberra Nature Map can give an overview of what you might find if you cared to go looking for them. The categories and species provided there have the expert oversight of Canberra’s Heino Lepp, who has provided detailed commentaries on many of them. For any curious naturalist, fungi provide a fascinating arena of exploration.
Main references:
- https://ucmp.berkeley.edu/fungi/fungilh.html
- https://hort.extension.wisc.edu/articles/mycorrhizae/
- https://canberra.naturemapr.org/categories/1696
- https://www.anbg.gov.au/fungi/mycorrhiza.html
Update on Yarramundi Trial
John Fitz Gerald
The last newsletter issue included details of two plots at Yarramundi Grassland where NCA sowed sterile Ryecorn.
The crop is growing well in the cold as shown in this photo (above left) of bright green leaves beside the newly resurfaced cycle path. This is in marked contrast to the mostly native grassland in the background, cloaked in drab winter colour, which remains short after its biomass reduction burn in September 2024. A second photo (above right) shows an early inflorescence starting to open.
A highlight for me when I poked around was to find some remnant native plants in the trial plots.
My final photo (left) is one (of five) Blue Devil which must have been dry enough to resist the herbicide applied in March; the green stems behind it are all Ryecorns. I also noticed a couple of plants of Bulbine Lily and Tricoryne sprouting nearby.
[Editor’s note: A related article by John ‘2025 Revegetation Trial with Ryecorn at Yarramundi Grassland’ was included in the May-June issue of this newsletter]
FOG’s Queanbeyan Adventure
Margaret Ning
On Sunday morning 25 May, a group of eleven gathered for our planned walks over the 2.2 hectare Queanbeyan Nature Reserve (QNR) near the Queanbeyan Racecourse and later, a short walk along the Queanbeyan River upstream of Dane Street. Even the weather was raring to go!
Our leaders, Tom Baker and Bill Willis of Queanbeyan Landcare, gave us extensive background information on native grasslands in the area, including the large triangular-shaped Letchworth Nature Reserve along the western side of Lanyon Drive, and the wildlife-rich, privately-owned property 'The Poplars' on the eastern side of Lanyon Drive. Both these reserves have colonies of the rare and endangered Rutidosis leptorrhynchoides, the Button Wrinklewort daisy.
There have also been past reports from both properties of the Canberra Grassland Earless Dragon. The Jerrabomberra Valley Grassland reserves created in 2004 nearby in the ACT also provide habitat for the Button Wrinklewort and the Earless Dragon. So, essentially the whole extended area of native grasslands (and the usual weeds) contains patches of Button Wrinklewort, all having a level of protection.
Queanbeyan NR_landscape at entry off Furlong Rd, Photo: Andrew Zelnik
Dedicated in 1989, QNR is a lovely shady grassy woodland reserve, with a nice level track through it and a healthy, stable population of the Button Wrinklewort at the western end. The occasional Button Wrinklewort was still flowering in the nature reserve, which was great to see. We spent an hour walking through what is an ecotone, (where the woodland grades into natural grassland*), all the while listening to a history of conservation and development in the area over nearly 40 years. Tom and Bill were full of fascinating anecdotes. The 1998
Management Plan for the QNR is currently under revision. Some isolated invasive plants were noted, mainly St John's wort and African lovegrass.
Returning to our vehicles, we moved across town to Dane Street on the Queanbeyan River, where we were given a briefing on that site’s management history. There has been a management plan for the river corridor since 1998, and it was the site of willow removal and native plantings around 1999-2001. Then from 2009-2011 two crews of Indigenous people were employed, along with contractors, to remove the remaining willows, and thousands of native plants were planted. Currently there is a bit of mystery about what the next step will be, as a $2m Council grant is on the table to remove weeds, do more plantings and publicise the platypus and rakali that inhabit the river - but the broader details are as yet unknown.
Queanbeyan River corridor, Dane St. Photo: Andrew Zelnik
We set off for a brisk 1km walk upstream towards Fairlane Estate, as Tom and Bill continued to point out examples of the various Landcare works that have occurred there. The Q&A continued. It was a very pleasant walk, with vegetation on both sides of the river, the possibility of platypus and rakali sightings, and knowing that the bird list for the walk area is almost 130 native species (link here ). Sadly we didn’t see any platypus, although we were almost at the right place at the right time on one occasion.
Thank you Tom and Bill, for showing our group of keen visitors around a couple of your local patches.
*Ecotones are transition areas where ecological communities, ecosystems and biotic regions coincide. These zones are rich in biodiversity, acting as vibrant meeting grounds between distinct ecosystems. For birds and animals the intertwined habitats provide critical space for courtship, nesting and foraging.
Three ‘natives’: close-up
John Fitz Gerald
Back to common natives this time, one daisy, one forb, one succulent.
My first daisy probably grows somewhere on every FoG work site – Chrysocephalum apiculatum or Common Everlasting. The species is a native perennial and occurs in all Australian states and territories but mostly across our country’s south. The ALA shows 27,500 records, over half in NSW but many in Victoria also. ACT has 1040 of them. I was surprised when I checked that the species’ inclusion will be a first for my close-ups. I am surprised no longer – the complexities of its subspecies have required me to search many information sources. My crash course started last year when I was very fortunate to be in the audience for an excellent presentation by Shelley Rowntree at the ANBG on names and definitions within this species. Shelley is undertaking a PhD at the University of New England, Armidale, working on a collection of DNA sequence data and taxonomy to analyse the C. apiculatum complex, which includes C. semipapposum. Shelley is basing her analyses on the complete study by Paul G. Wilson published in 2016. Wilson was a highly-esteemed botanist at the Herbarium of Western Australia and covered the country in this task, presenting a taxonomy-based key for continent-wide ID in his landmark paper, with detailed descriptions of 29 subspecies and framing the concept of a C. apiculatum complex. Wilson encountered systemic complexity: “Each subspecies grades into one or more of those subspecies whose natural distribution overlaps, or evidently once overlapped”; in other words, characteristics can be transitional between spatially-neighbouring subspecies. I searched using ALA to find that the Southern Tablelands division of NSW has 7 of these subspecies, all confirmed from preserved specimens and formally identified by Wilson. 32 preserved specimens from the ACT were also ID-ed by Wilson as belonging to 6 of the 7 subspecies from surrounding NSW. Two of these appear in the ACT Plant Census’ current version 4.1.
My image alongside shows 6 dark brown seeds from a tall plant in Canberra collected from a strip revegetated with native grasses and forbs (from regionally-sourced seed) on a trunk- sewer site in the suburb of Flynn. I trimmed the pappus bristles from 5 of the seeds leaving the bristles on the 6th seed that are barbed, becoming yellow and plumose at the tips.
The seeds are long and almost cylindrical. The scale bar in this image is 0.5mm long. For what it is worth, I see that Wilson’s key for ssp. variabile notes 4-7 pappus bristles as a characteristic, like that in my image, but I cannot confirm the full vegetative characters needed for definitive ssp. ID.
My second forb is Epilobium billardiereanum or Willowherb, a native perennial in both Australia and New Zealand. ALA lists 12,700 records, concentrating in states in the SE corner; ACT is credited with almost 550 records. Again, this plant is likely to be seen in the grasslands of all FoG work sites and has distinctively 4-petalled pink to white flowers. The fruit formed from flowering is erect, slender and cylindrical. All these aspects are well-illustrated in resources such as CNM. When ripe, each fruit splits wide open from the apex to release small seeds embedded in a fine silky fluff, aggregated from a coma attached to each seed. The fluff disperses easily in any wind and the small seed (right) detaches readily from it. Seeds are obovoid, light coloured and patterned by rows of tiny bumps. The scale bar here is 0.2mm.
My succulent is Portulaca oleracea or Purslane. It is also known by an indigenous name Munyeroo. This is a low-growing annual which can be a short-lived perennial, common in disturbed and cultivated sites. ALA shows 10,600 records from across Australia, 90 from the ACT. The status of this species is difficult to pin down with inconsistency between different sources - this is why ‘native’ is written between quotes in the title for this article.
Plants of the World Online (POWO) insists that the species is native only to most of N Africa, Europe and Asia as far east as Pakistan; elsewhere in its worldwide distribution the species has been introduced. Bean (2007) assessed the species to be partly native in Australia on the basis of his indigenous-plant system and suggested that the plant reached our shores before the British explorers visited the east coast.
Plantnet’s species map shows most NSW occurrences as native, others naturalised. Flora of NT considers the species to be native there. WA Florabase suggests that plants in the NW of the state (Kimberley) are native, elsewhere introduced. CNM sensibly avoids the argument by labelling the species 'cosmopolitan'. 5 seeds are shown in my image. These hard black seeds are small, partly flattened and densely covered with tiny protuberances. The scale bar here is 0.2mm. Some literature suggests that a vigorous plant can produce ten thousand seeds in a good season.
High magnification images were taken using the Nikon microscope at the National Seedbank in the Australian National Botanic Gardens. Images can be reproduced freely with reference to the Creative Commons licence CC BY.
Information above was gathered from websites, principally:
- ALA - www.ala.gov.au
- Wilson P.G. (2016) “A taxonomic treatment of Chrysocephalum apiculatum and C. semipapposum (Asteraceae: Gnaphalieae)”, Nuytsia, 27:33-73
- CNM - canberra.naturemapr.org plantnet.rbgsyd.nsw.gov.au
- Plants Of the World Online – powo.science.kew.org
- Flora Northern Territory online – eflora.nt.gov.au
- Bean A.R. (2007) “A new system for determining which plants are indigenous to Australia”, Australian Systematic Botany, 20, 1-43.
A note from webmaster Richard Bomford
Old versions of the FoG website are routinely archived by the National Library: https://trove.nla.gov.au/search/category/websites?keyword=fog&l-site=fog.org.au. We don’t delete much from the website, just keep adding, so old copies are generally just a subset of the current site, but it is good to know that it’s being safely put away in long term storage!
Hidingg in the grass: uncovering Melichrus (Ericaceae) species diversity in grassy ecosystems
Helen Kennedy, formerly University of New England, Armidale NSW.
Introduction by Andrew Zelnik:
In 2019 Helen was awarded a Friends of Grasslands (FOG) Grassy Ecosystem Grant of $1,500 to support her research into the diversity of Melichrus R.Br. in grassy ecosystems in eastern Australia. This research was part of her PhD at UNE which aimed to revise the taxonomy of all eastern Melichrus. This was off the back of her extensive field sampling and analysis of morphological (i.e. concerning physical appearance and structure) and genetic data to comprehensively describe species diversity, to explore evolutionary history, and to provide interactive identification tools. This report was originally submitted by Helen in the latter half of 2022 as part of her grant acquittal and is still current. It presents the major findings of her research as they relate to FOG’s modest grant contribution which enabled DNA analysis of samples from an additional 10 populations of Melichrus species needed to represent the Southern Tableland of NSW and ACT grassy ecosystems component within the larger eastern Australia project study area. The majority of FOG’s grant contributed to the related costs of genome-skimming Next Generation Sequencing at the Australian Genome Research Facility (AGRF). In 2024 Helen received her PhD from UNE. She currently works at the Australian National Herbarium, Centre for Australian National Biodiversity Research in Canberra. In addition to her 2020 research paper (see references below) Helen, again as lead author, has this year had another research paper published related to her PhD research, in Australian Systematic Botany (Vol 38 Article SB24031). It covers her entire eastern Australia study area and presents new lines of evidence which form the basis of detailed recommendations for a revised species taxonomy of Melichrus, including the description of eight new species, re-circumscription of previously described species, and the correction of a longstanding nomenclatural misapplication. FOG is kindly included in the acknowledgements.
Melichrus R.Br. (Ericaceae: Epacridoideae) is a genus of shrubs endemic to Australia. R.Br. in the nomenclature refers to the standard author abbreviation for Scottish botanist and paleobotanist, Robert Brown, who conducted botanic research including naming of many plant genera in Australia in the very early 1800s. The taxonomy currently consists of six described species (Paterson 1957; Kennedy et al. 2020), all confined to eastern Australia. In addition, six phrase-named species are accepted by the Australian Plant Census (APC; CHAH, accessed 23 May 2020), three in eastern Australia and three in southern Western Australia. A phrase-name is an informal name given to a plant taxon that has not yet been given a formal scientific name e.g. Melichrus sp. Inglewood (A.R.Bean 1652) Qld Herbarium).
The eastern Australian APC-listed taxa occur in Queensland (6 species), New South Wales (6), Australian Capital Territory (1) and Victoria (1) and are distributed from the coast to drier regions well inland (Fig. 1). Melichrus from Western Australia and the eastern states form sister clades (Powell et al. 1997; Puente- Lelièvre et al. 2016), allowing this study to focus on the eastern Australian Melichrus. Herein, unless otherwise specified, ‘Melichrus’ refers to the eastern clade. A clade is a monophyletic group or natural group of organisms composed of a common ancestor and all of its descendants. The majority of herbarium records of Melichrus are for grassy ecosystems, and most of them have been identified as M. urceolatus, common across south east Australia.
Fig 1: Map of all herbarium collection sites of Melichrus available through the Australian Virtual Herbarium.
Melichrus has received very little taxonomic study and its few treating authors have largely disagreed on species delimitation. Since the genus was last revised by Betsy Jackes (née Paterson) in 1957, several putative new species have been proposed. The aim of my PhD was to revise the genus Melichrus using modern methods to resolve problematic species boundaries, test putative new species and produce a biologically meaningful species taxonomy.
Throughout the first two years of my PhD I devoted considerable time to visiting herbaria and collecting in the field the plant material required to test species delimitation. During this time, I also hypothesised a new taxonomic structure for Melichrus, which I expressed as phrase-named putative species. I then used the material I had collected to test these hypotheses against the existing taxonomy using both morphological and molecular analyses.
To test species delimitation, numerous morphological characters were explored, photographed and recorded. This large dataset of morphological traits, was used in statistical and phenetic analyses.
The $1,500 grant from FOG made a substantial contribution to the molecular analyses used in this study. Several hundred individual plants were sampled across eastern Australia for inclusion in a next generation sequencing Diversity Arrays Technology (DArTseq) analysis. FOG’s grant funded the inclusion of almost 40 of these samples, which focussed on Melichrus populations in Southern Tablelands grasslands and grassy- woodland ecosystems in NSW and the ACT. This analysis produced an information rich dataset, of nearly 80,000 single nucleotide polymorphisms (SNPs). A stringently filtered final dataset was used in multiple analyses to examine patterns of relatedness and diversity including Principle coordinate analysis (PCoA), STRUCTURE, various disparity and diversity parameters and phylogenetic inference. Phylogenetics is the study of phylogenetic trees. A phylogenetic tree, phylogeny, or evolutionary tree is a branching diagram or tree showing the evolutionary relationships among various biological species or other entities based upon
similarities and differences in their physical or genetic characteristics. Here I won’t be sharing all of the results of this study, but instead will focus on a key taxonomic result which the FOG grant specifically contributed to.
Early observations made during fieldwork, herbaria visits and while reviewing the literature all indicated that several populations attributed to Melichrus urceolatus occurring in grassy ecosystems in New South Wales and Victoria may represent an undescribed species (Fig.2). I gave this putative species the phrase name Melichrus sp. Galambary, as I first collected it on Black Mountain (also known as Mount Galambary) in the Australian Capital Territory.
Morphological analyses provided evidence in support of the putative new species, segregated from Melichrus urceolatus. Melichrus sp. Galambary differs from M. urceolatus s.s. (sensu stricto – 'in the strict sense') in several vegetative and floral characters. A full morphological description of both species will be submitted for publication shortly and a species profile made available through the Flora of Australia.
Fig 2: Map of sampled populations of M. urceolatus (red) and M. sp. Galambary (blue) included in analyses.
The DArTseq analysis provided strong support for the proposed species status of M. sp. Galambary. Figure three displays PCoA results for the four axes which explain the largest amount of the variation in the genetic dataset. The ordination shows populations belonging to M. sp. Galambary (Fig. 2: blue) and M. urceolatus s.s (Fig. 2: red) cluster discreetly. All other taxa are in grey. Ordination (or gradient analysis), is a method of statistical analysis, comprising a suite of techniques, used mainly in exploratory data analysis. In the ordination space, quantities that are near each other share attributes (i.e. are similar to some degree), and dissimilar objects are farther from each other. Such relationships between the objects, on each of several axes or latent variables, are then characterised numerically and/or graphically in a two-dimensional plot.
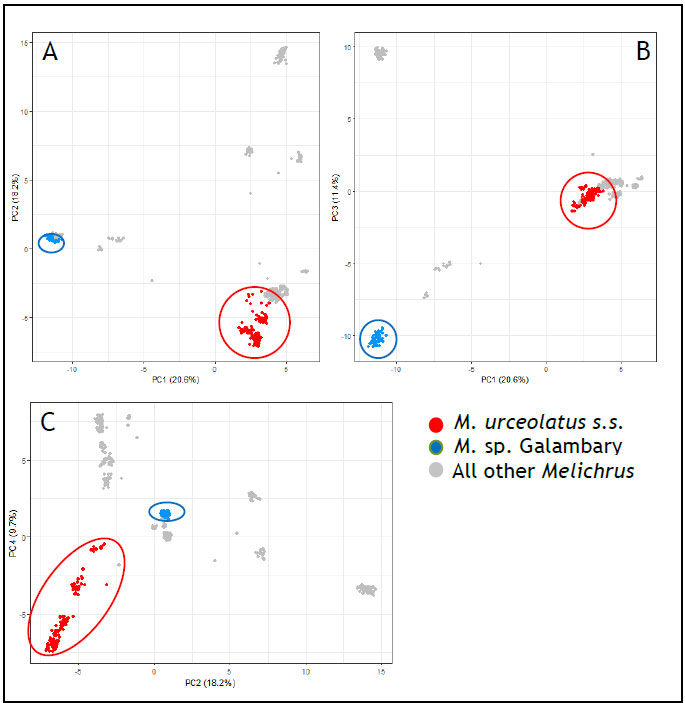
Fig 3: PCoA results for a DArTseq dataset representing the full morphological and geographical diversity of the genus Melichrus. Highlighted in red are all M. urceolatus s.s. samples. Highlighted in blue are
M. sp. Galambary samples. All other Melichrus taxa are shown in grey. A: PCoA axes 1 and 2, B: PCoA axes 1 and 3, C: PCoA axes 2 and 4. Phylogenetic analyses, using both the DArTseq dataset and a completely
independent molecular dataset showed that M. urceolatus and M. sp. Galambary represent distinct phylogenetic clades (Fig.4). The phylogeny shown in figure four was produced using a target capture DNA sequence dataset and inferred using ASTRALIII. The relationships seen in this phylogeny are mirrored in a maximum-likelihood tree produced using the DArTseq dataset. They provide strong support for M. sp.
Galambary as a monophyletic species (taxon consisting of a common ancestor and all its lineal descendants).
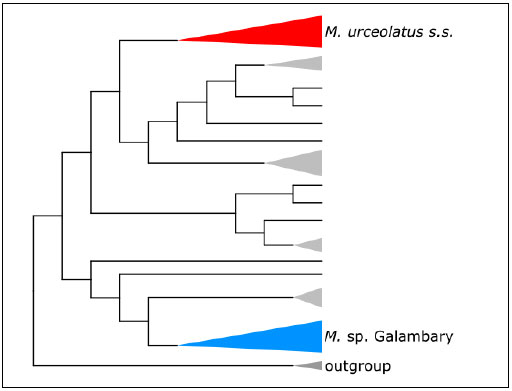
Fig 4: An ASTRALIII phylogeny for the genus Melichrus, inferred using a target capture DNA sequence dataset. Highlighting and labelling demonstrate the distinct phylogenetic clades that M. urceolatus s.s. (red) and M. sp. Galambary (blue) represent. Terminal nodes shown in black and grey represent other Melichrus taxa.
Considered together, the morphological and molecular evidence supports the proposed status of M. sp. Galambary as a species found predominantly in grassy woodlands across central and southern NSW, ACT and Victoria. I wanted to take this opportunity to highlight this result especially for members of Friends of Grasslands as this taxonomic change will affect vegetation descriptions and species inventories in grassy ecosystems in NSW, Victoria and the ACT. The full results of my thesis, including species descriptions and identification resources will be submitted for publication in scientific journals.
I would like to sincerely thank Friends of Grasslands for the significant support they have provided to this project. I would especially like to thank members Katerina Ng, Sue McIntyre and Maartje Sevenster for permission and assistance collecting Melichrus urceolatus s.s. on their properties and Andrew Zelnik for his administrative support.
References
Kennedy, HT, Telford, IRH, Crayn, DM, Bruhl, JJ (2020) Validation of two informally named species of Melichrus (Ericaceae: Epacridoideae) from north-eastern New South Wales. Telopea 23, 187-196.
Paterson, BR (1957) Revision of the genus Melichrus R.Br. (Epacridaceae). Proceedings of the Linnean Society of New South Wales. 82, 303-313.
Powell, JM, Morrison, DA, Gadek, PA, Crayn, DM, Quinn, CJ (1997) Relationships and Generic Concepts within Styphelieae (Epacridaceae). Australian Systematic Botany 10, 15-29.
Puente-Lelièvre, C, Hislop, M, Harrington, M, Brown, EA, Kuzmina, M, Crayn, DM (2016) A five-marker molecular phylogeny of the Styphelieae (Epacridoideae, Ericaceae) supports a broad concept of Styphelia. Australian Systematic Botany 28, 368-387.
The Mulloon Institute, and a search for NTG
Ann Milligan and Margaret Ning
The part of Mulloon Home Farm we visited on Friday afternoon 20 June is not only beautiful but also a rich example of natural temperate grassland (NTG). Our group’s attention was engaged for over 2 hours as we wandered across the valley sides and hilltops, spotting plant species until the sun sank into the western hills.
We were welcomed and accompanied by Peter Hazell and his three offsiders (Chris, Louis and Colby) who manage the property, its vegetation, livestock, natural assets and science. After acknowledging Traditional Owners and Elders, Peter explained the importance of bringing this land (at the top of the Great Dividing Range locally) into excellent ecological condition. First, it is significant to local Indigenous groups. Second, it is in the mid-reaches of the Mulloon Creek catchment (part of the Shoalhaven River catchment) and is a demonstration area for natural rehydration of agricultural catchments.
Around 2006 the owner, the late Tony Coote, decided to test Natural Sequence Farming – a system of managing catchment water flows devised by a NSW farmer named Peter Andrews – which consists of the partial damming of natural gullies and small creeks such as occurs when, say, rocks and branches accumulate in one place. This system of leaky weirs aims to imitate the chains of ponds that used to occur naturally along creeks before Europeans started clearing land for agriculture. Often those natural ponds would stay bank-full even when the creek had long since ceased to flow, because the ponds were connected to underlying floodplain aquifers. This helped keep the surroundings alive.
The Mulloon farms and all the landholders along the Mulloon Creek are installing leaky weirs now to slow the flow of water that would otherwise drain quickly off these upland water catchments after rainfall. The resulting ponding and slower flow keep the banks above the gullies wetter (rehydrated) and better able to maintain their plant cover. Monitoring of stream flow since 2006 suggests there is no apparent
detriment to the landholders downstream of Mulloon Creek along the water’s journey to the Shoalhaven.
Our group walked slowly, heads bent, cameras clicking, across the large paddocks on the hillside and hilltop and valley bottom (the subcatchment) exploring the numerous grassland species. There is a west- facing hillside stand of snow gums (Eucalyptus pauciflora), and a hilltop stand of E. mannifera, and remnant very old E. dives. There are expanses of pinkish Themeda (middle photo above) on the slopes and ridges, easily seen from a distance by the colour. Grasses on the eastern facing slope include Dichelachne. Beyond the creek at the valley bottom is a green and orange dirt heap remaining from former copper mining.
The subcatchment we explored is being considered by the NSW Biodiversity Conservation Trust for possible funding to support its management as grazed natural grassland and woodland, Peter said.
The main aim of our visit was to determine if the area we explored was in fact NTG, and it didn’t take us long to compile a plant list of just under 70 species to confirm that. In our group we had a keen lichen enthusiast who assured us that we would also have sighted a minimum of 20 lichen species (photo on the right above) during the course of the afternoon.
And Peter told us that a bioblitz-type week is planned for the coming spring. It will include soil sampling as well as days focusing on vegetation and fauna. We hope FOG will be able to join in.
Thank you, Peter and team, for welcoming us and for suggesting we come back sooner rather than later!
All photos in this article are by Ann Milligan
Advocacy Report
Matt Whitting
June
Letter to the Prime Minister about necessary environmental law reform, 10/6
FOG President Jamie Pittock signed on to a letter by members of the ‘Places You Love Alliance’ calling on the PM to strengthen Australia’s nature protection laws.
Strategic Bushfire Management Plan (v5) (ACT), 10/6
FOG commented on Version 5 of the Strategic Bushfire Management Plan. We:
- Suggested ways of thinking about burn frequencies;
- Argued asset protection zones should be within (not beyond) development areas; and
- Put recommendations for the coordination and involvement of First Nations people in burn management processes.
Comment on the Environment Impact Statement (EIS) for the Upper and Lower Tumut 330 kV Transmission Line Realignment (EIS202300023), 10/6
FOG commented on the above EIS noting it did not present actual mitigation measures as required by the EIS Scoping Document. We said, as a result, that it was not possible to assess the impacts of the proposal.
Comment on landscape plans for the Drake Brockman Drive road reserve, 6/6
John Fitz Gerald & Ken Hodgkinson commented on the Ginninderry Joint Venture’s landscape plans for the Drake Brockman Drive Road Reserve; in particular, its plant schedule.
Comment on unpublished draft of the National Recovery Plan for Golden Sun Moth, 3/6
FOG provided comment on an unpublished version of the draft plan. Another opportunity will be provided to review this plan when the draft is published and public comment is invited.
May
Meeting with Minister Cheyne, ACT Minister for City Services, 28/5
Sarah Sharp, Julia Raine and Matt Whitting from FOG met with the Minister for Transport and City Services (TCCS), the Hon Tara Cheyne, to discuss achieving greater coordination of land management in the ACT. Sarah used a map produced for FOG to identify the urban areas, outside of reserves, managed by officers in her portfolio, that are of outstanding conservation value. We suggested drawing experts from across the ACT Government into a Bush Regeneration Crew, or similar, to lead in the better and more consistent management of these outstanding areas and all other unleased areas in ways that will maximise nature’s chances of surviving and thriving.
Meeting with Alicia Payne, MP for the Federal electorate of Canberra, 27/5
Julia Raine and Matt Whitting from FOG, and Simon Copland from the Conservation Council, met the Federal Member for Canberra, the Hon Alicia Payne, to discuss the northern road between Fairbairn Avenue and Majura Road in Pialligo. We outlined our views about the construction of the road by Canberra Airport Group (CAG), stressing our concern about the unnecessary loss of natural temperate grassland that is habitat for the Canberra Grassland Earless Dragon (CGED). We also outlined what is needed to achieve good conservation outcomes throughout the Majura Valley, including by avoiding further loss of/development in the grasslands. We requested and Alicia agreed, wherever and whenever she could, to seek the cooperation of ministers responsible, and of the managers of public and private land in the Majura Valley, to work toward the cooperative management of the Majura Valley for the better protection of the area’s nature.
Meeting with members of the Greens Party, ACT level, 23/5
Greens members Shane Rattenbury and Jo Clay invited Jamie Pittock and Matt Whitting from FOG to provide a review of our meeting with the ACT Minister for the Environment the previous day.
Meeting with Minister Orr, ACT Minister for the Environment 22/5
The ACT Minister for the Environment Suzanne Orr welcomed Jamie Pittock and Matt Whitting from FOG and Michael Thomson and Zarko Danilov from CAG to a meeting to discuss the northern road between Fairbairn Avenue and Majura Road in Pialligo. Jamie outlined FOG’s concerns, including that FOG is concerned about the decline in biodiversity values that has occurred under CAG’s management on the land it has leased for 25 years, and Michael responded.
Ultimately, talk turned to what FOG wants next which is:
- Transparency from CAG about how they will achieve the outcomes they are advocating for the CGED and for Natural Temperate Grassland (NTG);
- Greater contribution to recovery efforts and research; and
- Cooperation (if not leadership) from CAG on management of CGED and NTG, including in regional efforts to, for example, manage threats across tenures.
Letter to Minister Orr, 22/5
On 22 May, Jamie Pittock wrote to the Minister: expressing deep concern regarding the ongoing loss and degradation of Natural Temperate Grassland within the ACT; and calling on her to mandate an assessment by the Commissioner for Sustainability and the Environment of the extent of habitat loss and degradation that is affecting the Canberra Dragons, including to identify how best to protect and restore its remaining habitat across land tenures.
Complaint to the Commissioner for Sustainability and Environment, 21/5
Citing information included in a report on implementation of the ACT Nature Conservation Strategy 2013 – 2023, FOG complained that development and neglect is destroying and degrading lowland areas in the ACT. Between 2019 and 2023, 882 ha of remnant lowland vegetation was lost, a massive 684 ha or 78 per cent of which was or could have been listed as one of two threatened ecological communities - NTG (17 ha) and Box Gum Woodlands (667 ha). The complaint went on to explain that not enough is planned or being done to protect the habitat of the CGED.
Letter to TCCS concerning their review of the Public Unleased Land Act 2013 (ACT), 15/5
On 3 April, TCCS had briefed the Conservation Council on their review of the above Act. Matt Whitting attended as a volunteer assisting the Council. In turn, on 15 May, FOG and the Council wrote to TCCS to outline the importance of ensuring biodiversity values within and outside reserves are retained when authorisations are granted under the Act for the use of public unleased land.
Letter to the new Environment Minister on the northern road at Canberra Airport, 13/5
On 13 May, the Hon Murray Watt MP was sworn in as the new federal environment minister. FOG and the Conservation Council wrote to him that morning calling for urgent action to revoke the approval
granted to CAG for the northern road at Canberra Airport.
Media release responding to the release of the Action Plan for Dragons and funding for implementation of the Plan, 8/5
On 7 May, the ACT Government released its Action Plan for CGED and announced funding to implement it. On 8 May, FOG’s representative on the group that helped to develop the Plan, Geoff Robertson, was notified. Two media stories mention these developments and the protest on 6 May: The Canberra Times ‘Big boost for breeding program saving Canberra’s cryptic little beasts’ and ABC News ‘The [Dragon] is fighting for survival amid construction of a new airport road’.
Attendance at protest to save Canberra Dragons, 6/5
On 6 May, FOG members attended a Snap Protest organised by the Conservation Council ACT Region aimed at raising awareness about the plight of Canberra Dragons.
Contact us
|
General inquiries, health and safety |
|
|
Media inquiries |
0407265131 (Jamie Pittock), 0403221117 (Geoff Robertson) |
|
Membership enquiries, |
|
|
Events & work parties |
|
|
Book order forms |
|
|
Small grassy ecosystem grants |
|
|
Advocacy contact |
|
|
Website matters |
|
|
Projects / work party contacts |
Hall Cemetery, Ginninderry: john.fitzgerald@fog.org.au. Scrivener’s Hut, Gurubung Dhaura (Stirling Park), Blue Gum Point & Yarramundi Grassland: jamie.pittock@fog.org.au. Budjan Galindji (Franklin) Grasslands, TSRs & Old Cooma Common: margaret.ning@fog.org.au. Scottsdale: |
|
Newsletter contact |
|
|
Contact addresses |
|
|
Payments & accounts |
|
|
Annual reports |
|
|
FOG Committee |
Photo: February 2025, FOG work party in action in "our" patch in the southeast corner of the 20 ha Budjan Galindji Nature Reserve, Franklin ACT. In the foreground an abundance of flowering native Lemon Beauty Heads (Calocephalus citreus), Kangaroo Grass and Speargrass, primarily from assisted natural regeneration through weed control enhancing the condition, both in terms of native plant species abundance and diversity, of this 1.6 ha remnant patch of critically endangered Natural Temperate Grassland. It is also habitat for threatened fauna species including the Striped Legless Lizard, Golden Sun Moth and Perunga Grasshopper. This provides a payoff for FOG's involvement, particularly since 2020 through its volunteering efforts including regular work parties and a good working relationship with ACT Parks and Conservation management and staff. Photo and caption: Andrew Zelnik.
News of Friends of Grasslands
is published by
Friends of Grasslands Inc.
PO Box 440, Jamison Centre
ACT 2614
